CSF progranulin increases in the course of Alzheimer's disease and is associated with sTREM2, neurodegeneration and cognitive decline
EMBO Mol Med. 2018 Dec;10(12). pii: e9712.
| Authors/Editors: |
Marc Suárez-Calvet Anja Capell Miguel Ángel Araque Caballero Estrella Morenas-Rodríguez Katrin Fellerer Nicolai Franzmeier Gernot Kleinberger Erden Eren Yuetiva Deming Laura Piccio Celeste M Karch Carlos Cruchaga Katrina Paumier Randall J Bateman Anne M Fagan John C Morris Johannes Levin Adrian Danek Mathias Jucker Colin L Masters Martin N Rossor John M Ringman Leslie M Shaw John Q Trojanowski Michael Weiner Michael Ewers Christian Haass for the Dominantly Inherited Alzheimer Network & for the Alzheimer's Disease Neuroimaging Initiative |
|---|---|
| Publication Date: | 2018 |
| Type of Publication: | Journal Article |
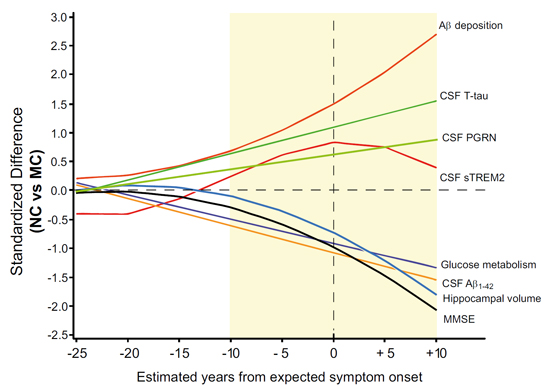
Neuroinflammation and microgliosis are key pathological features of Alzheimer's disease (AD). Cerebrospinal fluid (CSF) protein levels analysis in large AD patients' cohorts reveals that CSF levels of Progranulin (PGRN) together with soluble TREM2 may serve as a microglia activity marker in AD.
- CSF PGRN increases in carriers of autosomal‐dominant AD from the Dominant Inherited Alzheimer's Disease Network (DIAN), causing dominant mutations 10 years before the expected symptom onset.
- CSF PGRN increases in late‐onset AD patients from the Alzheimer's disease Neuroimaging Initiative (ADNI) cohort during the course of the disease and is associated with cognitive decline.
- CSF PGRN and CSF soluble TREM2 (sTREM2), both of which have key regulatory functions in microglia, are associated specifically when there is underlying neurodegeneration.
- CSF PGRN together with CSF sTREM2 may serve as microglial activity markers and could be used to prove target engagement in clinical trials aiming to modulate microglial activity.
Abstract
Progranulin (PGRN) is predominantly expressed by microglia in the brain, and genetic and experimental evidence suggests a critical role in Alzheimer’s disease (AD). We asked whether PGRN expression is changed in a disease severity-specific manner in AD. We measured PGRN in cerebrospinal fluid (CSF) in two of the best-characterized AD patient cohorts, namely the Dominant Inherited Alzheimer’s Disease Network (DIAN) and the Alzheimer’s Disease Neuroimaging Initiative (ADNI). In carriers of AD causing dominant mutations, cross-sectionally assessed CSF PGRN increased over the course of the disease and significantly differed from non-carriers 10 years before the expected symptom onset. In late-onset AD, higher CSF PGRN was associated with more advanced disease stages and cognitive impairment. Higher CSF PGRN was associated with higher CSF soluble TREM2 (triggering receptor expressed on myeloid cells 2) only when there was underlying pathology, but not in controls. In conclusion, we demonstrate that, although CSF PGRN is not a diagnostic biomarker for AD, it may together with sTREM2 reflect microglial activation during the disease.



