Enhancing protective microglial activities with a dual function TREM2 antibody to the stalk region
EMBO Mol Med. 2020 Apr 7;12(4):e11227.
| Authors/Editors: |
Kai Schlepckow Kathryn M. Monroe Gernot Kleinberger Ludovico Cantuti-Castelvetri Samira Parhizkar Dan Xia Michael Willem Georg Werner Nadine Pettkus Bettina Brunner Alice Sülzen Brigitte Nuscher Heike Hampel Xianyuan Xiang Regina Feederle Sabina Tahirovic Joshua I. Park Rachel Prorok Cathal Mahon Chun-Chi Liang Ju Shi Do Jin Kim Hanna Sabelström Fen Huang Gilbert Di Paolo Mikael Simons Joseph W. Lewcock Christian Haass |
|---|---|
| Publication Date: | 2020 |
| Type of Publication: | Journal Article |
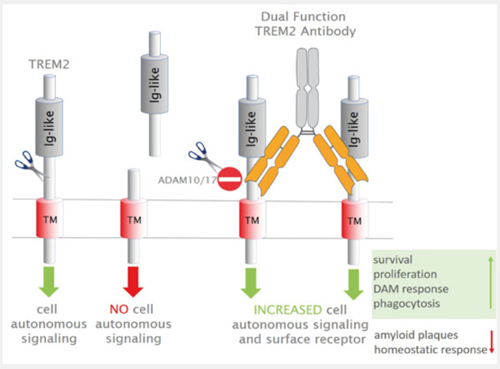
This study describes the discovery and characterization of a novel TREM2 antibody, which induces protective microglial functions and provides a basis for the development of human antibodies with a similar mechanistic profile for treatment of Alzheimer's disease.
- An antibody directed to the stalk region of TREM2 prevents its shedding and increases cell autonomous signaling.
- Addition of this TREM2 antibody to myeloid cells in vitro stimulates phagocytosis, and improves cell survival.
- TREM2 antibody treatment increases TREM2 expression on brain microglia, decreases homeostatic markers and reduces amyloid plaque pathology in a mouse model of Alzheimer's disease.
- Antibody mediated stimulation of TREM2 signaling may be efficacious in Alzheimer's disease as well as other neurodegenerative disorders and obesity‐associated metabolic syndromes.
Abstract:
Triggering receptor expressed on myeloid cells 2 (TREM2) is essential for the transition of homeostatic microglia to a disease‐associated microglial state. To enhance TREM2 activity, we sought to selectively increase the full‐length protein on the cell surface via reducing its proteolytic shedding by A Disintegrin And Metalloproteinase (i.e., α‐secretase) 10/17. We screened a panel of monoclonal antibodies against TREM2, with the aim to selectively compete for α‐secretase‐mediated shedding. Monoclonal antibody 4D9, which has a stalk region epitope close to the cleavage site, demonstrated dual mechanisms of action by stabilizing TREM2 on the cell surface and reducing its shedding, and concomitantly activating phospho‐SYK signaling. 4D9 stimulated survival of macrophages and increased microglial uptake of myelin debris and amyloid β‐peptide in vitro. In vivo target engagement was demonstrated in cerebrospinal fluid, where nearly all soluble TREM2 was 4D9‐bound. Moreover, in a mouse model for Alzheimer's disease‐related pathology, 4D9 reduced amyloidogenesis, enhanced microglial TREM2 expression, and reduced a homeostatic marker, suggesting a protective function by driving microglia toward a disease‐associated state.



