Loss of Bace2 in zebrafish affects melanocyte migration and is distinct from Bace1 knock out phenotypes
J Neurochem. 2013 Feb 14. doi: 10.1111/jnc.12198. [Epub ahead of print]
| Authors/Editors: |
van Bebber F Hruscha A Willem M Schmid B Haass C |
|---|---|
| Publication Date: | 2013 |
| Type of Publication: | Journal Article |
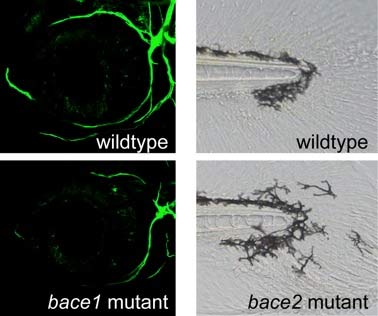
ABSTRACT:
Alzheimer's disease (AD) is the most frequent dementia. Pathologically, AD is characterized by the accumulation of senile plaques composed of amyloid β-peptide (Aβ). Two proteases, β- and γ-secretase proteolytically generate Aβ from its precursor, the ß-amyloid precursor protein (APP). Inhibition of β- secretase, also referred to as beta-site APP cleaving enzyme (BACE1) or γ-secretase is therefore of prime interest for the development of amyloid lowering drugs. To assess the in vivo function of zebrafish Bace1 (zBace1) we generated zBace1 knock out fish by zinc finger nuclease mediated genome editing. bace1 mutants (bace1 -/-) are hypomyelinated in the PNS while the CNS is not affected. Moreover the number of mechanosensory neuromasts is elevated in bace1 -/-. Mutations in zebrafish Bace2 (zBace2) revealed a distinct melanocyte migration phenotype, which is not observed in bace1 -/-. Double homozygous bace1 -/-; bace2 -/- fish do not enhance the single mutant phenotypes indicating non-redundant distinct physiological functions. Single homozygous bace1 mutants as well as double homozygous bace1 and bace2 mutants are viable and fertile suggesting that Bace1 is a promising drug target without major side effects. The identification of a specific bace2 -/- associated phenotype further allows improving selective Bace1 inhibitors and to distinguish between Bace 1 and Bace 2 inhibition in vivo. © 2013 International Society for Neurochemistry, J. Neurochem. (2013) 10.1111/jnc.12198.



