Monomethylated and unmethylated FUS exhibit increased binding to Transportin and distinguish FTLD-FUS from ALS-FUS
Acta Neuropathol. 2016 Apr;131(4):587-604
| Authors/Editors: |
Marc Suárez-Calvet Manuela Neumann Thomas Arzberger Claudia Abou-Ajram Eva Funk Hannelore Hartmann Dieter Edbauer Elisabeth Kremmer Christoph Göbl Moritz Resch Benjamin Bourgeois Tobias Madl Stefan Reber Daniel Jutzi Marc-David Ruepp Ian R. A. Mackenzie Olaf Ansorge Dorothee Dormann Christian Haass |
|---|---|
| Publication Date: | 2016 |
| Type of Publication: | Journal Article |
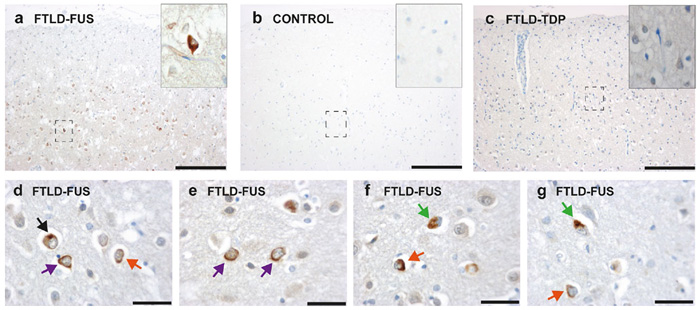
Deposition of the nuclear DNA/RNA-binding protein Fused in sarcoma (FUS) in cytosolic inclusions is a common hallmark of some cases of frontotemporal lobar degeneration (FTLD-FUS) and amyotrophic lateral sclerosis (ALS-FUS). Whether both diseases also share common pathological mechanisms is currently unclear. Based on our previous finding that FUS deposits are hypomethylated in FTLD-FUS but not in ALS-FUS, we have now investigated whether genetic or pharmacological inactivation of Protein arginine methyltransferase 1 (PRMT1) activity results in unmethylated FUS or in alternatively methylated forms of FUS. To do so, we generated FUS-specific monoclonal antibodies that specifically recognize unmethylated arginine (UMA), monomethylated arginine (MMA) or asymmetrically dimethylated arginine (ADMA). Loss of PRMT1 indeed not only results in an increase of UMA FUS and a decrease of ADMA FUS, but also in a significant increase of MMA FUS. Compared to ADMA FUS, UMA and MMA FUS exhibit much higher binding affinities to Transportin-1, the nuclear import receptor of FUS, as measured by pulldown assays and isothermal titration calorimetry. Moreover, we show that MMA FUS occurs exclusively in FTLD-FUS, but not in ALS-FUS. Our findings therefore provide additional evidence that FTLD-FUS and ALS-FUS are caused by distinct disease mechanisms although both share FUS deposits as a common denominator.