Content
Alzheimer research
Alzheimer Research
Alzheimer's disease (AD) is the most common cause of progressive cognitive decline in our ageing society. Pathologically, AD is characterized by the accumulation of senile plaques, which are predominantly composed of the amyloid β-peptide (Aβ).
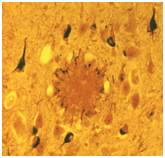
Figure 1: Amyloid plaque
Back in 1992, I discovered that Aβ is derived from the membrane bound beta-amyloid precursor protein (βAPP) by sequential proteolytic cleavage. β-secretase (BACE) and γ-secretase generate Aβ in a physiologically normal process. Mutations associated with familial Alzheimer's disease are key for our research, since they provide direct implications for the disease mechanisms.
We currently focus on multiple aspects of AD research including
- Regulation of BACE expression
- Alternative APP processing pathways
- Physiological function of APP processing derivatives
- Clearance of Aβ
- Microglial activation and function in Alzheimer's disease and other neurodegenerative disorders
- CSF markers in close collaboration with DIAN
Funding
European Research Council under the European Union’s Seventh Framework Program (FP7/2007–2013)
ERC grant agreement no. 321366-Amyloid (advanced grant)
MetLife Award for Medical Research
DFG Research Group 2290: Understanding Intramembrane Proteolysis
Further Reading
Alzheimer - Mechanismen und therapeutische Ansätze. Warum wir im Alter dement werden

